Target Information
| Target General Information | Top | |||||
|---|---|---|---|---|---|---|
| Target ID |
T12475
(Former ID: TTDS00273)
|
|||||
| Target Name |
Gonadotropin-releasing hormone receptor (GNRHR)
|
|||||
| Synonyms |
Hypothalamic gonadotropin-releasing hormone receptor; Gonadotrophin releasing hormone receptor; GnRH-R; GnRH receptor; GRHR
Click to Show/Hide
|
|||||
| Gene Name |
GNRHR
|
|||||
| Target Type |
Successful target
|
[1] | ||||
| Disease | [+] 6 Target-related Diseases | + | ||||
| 1 | Endometriosis [ICD-11: GA10] | |||||
| 2 | Ovarian developmental anomaly [ICD-11: LB45] | |||||
| 3 | Ovarian dysfunction [ICD-11: 5A80] | |||||
| 4 | Pituitary gland disorder [ICD-11: 5A60-5A61] | |||||
| 5 | Prostate cancer [ICD-11: 2C82] | |||||
| 6 | Solid tumour/cancer [ICD-11: 2A00-2F9Z] | |||||
| Function |
Receptor for gonadotropin releasing hormone (GnRH) that mediates the action of GnRH to stimulate the secretion of the gonadotropic hormones luteinizing hormone (LH) and follicle-stimulating hormone (FSH). This receptor mediates its action by association with G-proteins that activate a phosphatidylinositol-calcium second messenger system. Isoform 2 may act as an inhibitor of GnRH-R signaling.
Click to Show/Hide
|
|||||
| BioChemical Class |
GPCR rhodopsin
|
|||||
| UniProt ID | ||||||
| Sequence |
MANSASPEQNQNHCSAINNSIPLMQGNLPTLTLSGKIRVTVTFFLFLLSATFNASFLLKL
QKWTQKKEKGKKLSRMKLLLKHLTLANLLETLIVMPLDGMWNITVQWYAGELLCKVLSYL KLFSMYAPAFMMVVISLDRSLAITRPLALKSNSKVGQSMVGLAWILSSVFAGPQLYIFRM IHLADSSGQTKVFSQCVTHCSFSQWWHQAFYNFFTFSCLFIIPLFIMLICNAKIIFTLTR VLHQDPHELQLNQSKNNIPRARLKTLKMTVAFATSFTVCWTPYYVLGIWYWFDPEMLNRL SDPVNHFFFLFAFLNPCFDPLIYGYFSL Click to Show/Hide
|
|||||
| 3D Structure | Click to Show 3D Structure of This Target | AlphaFold | ||||
| HIT2.0 ID | T45C0Q | |||||
| Drugs and Modes of Action | Top | |||||
|---|---|---|---|---|---|---|
| Approved Drug(s) | [+] 8 Approved Drugs | + | ||||
| 1 | Abarelix | Drug Info | Approved | Prostate cancer | [5], [6] | |
| 2 | Cetrorelix | Drug Info | Approved | Ovarian stimulation | [7], [8] | |
| 3 | Ganirelix | Drug Info | Approved | Ovulation dysfunction | [9], [10] | |
| 4 | Gonadorelin | Drug Info | Approved | Hypothalamic hypogonadism | [10], [11] | |
| 5 | Leuprorelin acetate | Drug Info | Approved | Prostate cancer | [12] | |
| 6 | Nafarelin | Drug Info | Approved | Endometriosis | [10], [13] | |
| 7 | Relugolix | Drug Info | Approved | Prostate cancer | [14] | |
| 8 | Triptorelin | Drug Info | Approved | Solid tumour/cancer | [10], [15] | |
| Clinical Trial Drug(s) | [+] 7 Clinical Trial Drugs | + | ||||
| 1 | AEZS-108 | Drug Info | Phase 3 | Acute lymphoblastic leukaemia | [18] | |
| 2 | GnRH | Drug Info | Phase 3 | Female infertility | [19] | |
| 3 | ACYLINE | Drug Info | Phase 2 | Prostate cancer | [20] | |
| 4 | ASP-1707 | Drug Info | Phase 2 | Endometriosis | [21] | |
| 5 | KLH-2109 | Drug Info | Phase 2 | Endometriosis | [22] | |
| 6 | Ozarelix | Drug Info | Phase 2 | Prostate disease | [23] | |
| 7 | PEP-223/CoVaccine HT | Drug Info | Phase 1/2 | Prostate cancer | [25] | |
| Discontinued Drug(s) | [+] 7 Discontinued Drugs | + | ||||
| 1 | Antide | Drug Info | Discontinued in Phase 2 | Prostate hyperplasia | [27], [28] | |
| 2 | TAK-013 | Drug Info | Discontinued in Phase 2 | Uterine fibroids | [29], [30] | |
| 3 | A-75998 | Drug Info | Discontinued in Phase 1 | Hormone deficiency | [31] | |
| 4 | NBI-42902 | Drug Info | Discontinued in Phase 1 | Prostate cancer | [32], [33] | |
| 5 | Prostate cancer vaccine | Drug Info | Discontinued in Phase 1 | Prostate cancer | [34] | |
| 6 | Ramorelix | Drug Info | Discontinued in Phase 1 | Esophageal cancer | [35] | |
| 7 | D-26344 | Drug Info | Terminated | Solid tumour/cancer | [38] | |
| Preclinical Drug(s) | [+] 1 Preclinical Drugs | + | ||||
| 1 | 5-(N,N-hexamethylene)-amiloride | Drug Info | Preclinical | Coronavirus infection | [36] | |
| Mode of Action | [+] 5 Modes of Action | + | ||||
| Modulator | [+] 7 Modulator drugs | + | ||||
| 1 | Abarelix | Drug Info | [39] | |||
| 2 | Triptorelin | Drug Info | [45] | |||
| 3 | AEZS-108 | Drug Info | [46] | |||
| 4 | GnRH | Drug Info | [47] | |||
| 5 | Ozarelix | Drug Info | [23] | |||
| 6 | detirelix | Drug Info | [59], [60] | |||
| 7 | Org-30850 | Drug Info | [64] | |||
| Antagonist | [+] 13 Antagonist drugs | + | ||||
| 1 | Cetrorelix | Drug Info | [1], [40] | |||
| 2 | Ganirelix | Drug Info | [41] | |||
| 3 | Relugolix | Drug Info | [14] | |||
| 4 | ASP-1707 | Drug Info | [49] | |||
| 5 | Antide | Drug Info | [51] | |||
| 6 | A-75998 | Drug Info | [53] | |||
| 7 | Ramorelix | Drug Info | [55] | |||
| 8 | AEZS-115 | Drug Info | [47] | |||
| 9 | antarelix | Drug Info | [58] | |||
| 10 | furan-1 | Drug Info | [61] | |||
| 11 | IN-3 | Drug Info | [62] | |||
| 12 | LXT-101 | Drug Info | [63] | |||
| 13 | [125I]cetrorelix | Drug Info | [67] | |||
| Agonist | [+] 4 Agonist drugs | + | ||||
| 1 | Gonadorelin | Drug Info | [42] | |||
| 2 | Leuprorelin acetate | Drug Info | [43] | |||
| 3 | Nafarelin | Drug Info | [44] | |||
| 4 | KLH-2109 | Drug Info | [50] | |||
| Inhibitor | [+] 35 Inhibitor drugs | + | ||||
| 1 | ACYLINE | Drug Info | [48] | |||
| 2 | TAK-013 | Drug Info | [52] | |||
| 3 | NBI-42902 | Drug Info | [54] | |||
| 4 | D-26344 | Drug Info | [57] | |||
| 5 | AN-207 | Drug Info | [47] | |||
| 6 | AZALINE B | Drug Info | [48] | |||
| 7 | PGlu-His-Trp-Ser-His-D-Tyr-Leu-Arg-Pro-Gly-NH2 | Drug Info | [65] | |||
| 8 | PGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2 | Drug Info | [65] | |||
| 9 | T-98475 | Drug Info | [52] | |||
| 10 | WAY-207024 | Drug Info | [66] | |||
| 11 | [4Aph(CO-(C2H4O)3-C2H4-NH-CO-NH-OCH3)5]degarelix | Drug Info | [68] | |||
| 12 | [4Aph(CO-NH-(C2H4O)2-C2H5)5]degarelix | Drug Info | [68] | |||
| 13 | [4Aph(CO-NH-OCH3)5,D-4Aph(CO-NH-OCH3)6]degarelix | Drug Info | [68] | |||
| 14 | [4Aph(CO-NH-OCH3)5]degarelix | Drug Info | [68] | |||
| 15 | [4Aph(CO-NH-OH)5]degarelix | Drug Info | [68] | |||
| 16 | [D-4Aph(CO-NH-OCH3)6]degarelix | Drug Info | [68] | |||
| 17 | [D-4Aph(CO-NH-OH)6]degarelix | Drug Info | [68] | |||
| 18 | [D-Dab(CO-NH-OCH3)3]degarelix | Drug Info | [68] | |||
| 19 | [D-Dab(CO-NH-OH)3]degarelix | Drug Info | [68] | |||
| 20 | [D-Dap(CO-NH-OCH3)3]degarelix | Drug Info | [68] | |||
| 21 | [D-Dap(CO-NH-OH)3]degarelix | Drug Info | [68] | |||
| 22 | [D-Gln3,D-Ncy(isopropyl)7]acyline | Drug Info | [48] | |||
| 23 | [D-Gln3,Ncy(SO,isopropyl)7]acyline | Drug Info | [48] | |||
| 24 | [D-Ncy(isopropyl)7]acyline | Drug Info | [48] | |||
| 25 | [D-Ncy(methyl)10] acyline | Drug Info | [48] | |||
| 26 | [D-Ncy(methyl)4]acyline | Drug Info | [48] | |||
| 27 | [D-Ncy(SO,isopropyl)7]acyline | Drug Info | [48] | |||
| 28 | [D-Ncy(SO,methyl)10]acyline | Drug Info | [48] | |||
| 29 | [Ncy(2-naphthyl)1]acyline | Drug Info | [48] | |||
| 30 | [Ncy(isopropyl)7]acyline | Drug Info | [48] | |||
| 31 | [Ncy(methyl)10]acyline | Drug Info | [48] | |||
| 32 | [Ncy(methyl)4]acyline | Drug Info | [48] | |||
| 33 | [Ncy(SO,isopropyl)7]acyline | Drug Info | [48] | |||
| 34 | [Ncy(SO,methyl)10]acyline | Drug Info | [48] | |||
| 35 | [Ncy(SO,methyl)4]acyline | Drug Info | [48] | |||
| Modulator (allosteric modulator) | [+] 1 Modulator (allosteric modulator) drugs | + | ||||
| 1 | 5-(N,N-hexamethylene)-amiloride | Drug Info | [56] | |||
| Cell-based Target Expression Variations | Top | |||||
|---|---|---|---|---|---|---|
| Cell-based Target Expression Variations | ||||||
| Different Human System Profiles of Target | Top |
|---|---|
|
Human Similarity Proteins
of target is determined by comparing the sequence similarity of all human proteins with the target based on BLAST. The similarity proteins for a target are defined as the proteins with E-value < 0.005 and outside the protein families of the target.
A target that has fewer human similarity proteins outside its family is commonly regarded to possess a greater capacity to avoid undesired interactions and thus increase the possibility of finding successful drugs
(Brief Bioinform, 21: 649-662, 2020).
Human Pathway Affiliation
of target is determined by the life-essential pathways provided on KEGG database. The target-affiliated pathways were defined based on the following two criteria (a) the pathways of the studied target should be life-essential for both healthy individuals and patients, and (b) the studied target should occupy an upstream position in the pathways and therefore had the ability to regulate biological function.
Targets involved in a fewer pathways have greater likelihood to be successfully developed, while those associated with more human pathways increase the chance of undesirable interferences with other human processes
(Pharmacol Rev, 58: 259-279, 2006).
Biological Network Descriptors
of target is determined based on a human protein-protein interactions (PPI) network consisting of 9,309 proteins and 52,713 PPIs, which were with a high confidence score of ≥ 0.95 collected from STRING database.
The network properties of targets based on protein-protein interactions (PPIs) have been widely adopted for the assessment of target’s druggability. Proteins with high node degree tend to have a high impact on network function through multiple interactions, while proteins with high betweenness centrality are regarded to be central for communication in interaction networks and regulate the flow of signaling information
(Front Pharmacol, 9, 1245, 2018;
Curr Opin Struct Biol. 44:134-142, 2017).
Human Similarity Proteins
Human Pathway Affiliation
Biological Network Descriptors
|
|
| Protein Name | Pfam ID | Percentage of Identity (%) | E value |
|---|---|---|---|
| Olfactory receptor 2T34 (OR2T34) | 26.515 (35/132) | 3.94E-04 | |
| Olfactory receptor 2T3 (OR2T3) | 26.515 (35/132) | 5.73E-04 |
| KEGG Pathway | Pathway ID | Affiliated Target | Pathway Map |
|---|---|---|---|
| Neuroactive ligand-receptor interaction | hsa04080 | Affiliated Target |

|
| Class: Environmental Information Processing => Signaling molecules and interaction | Pathway Hierarchy | ||
| GnRH signaling pathway | hsa04912 | Affiliated Target |
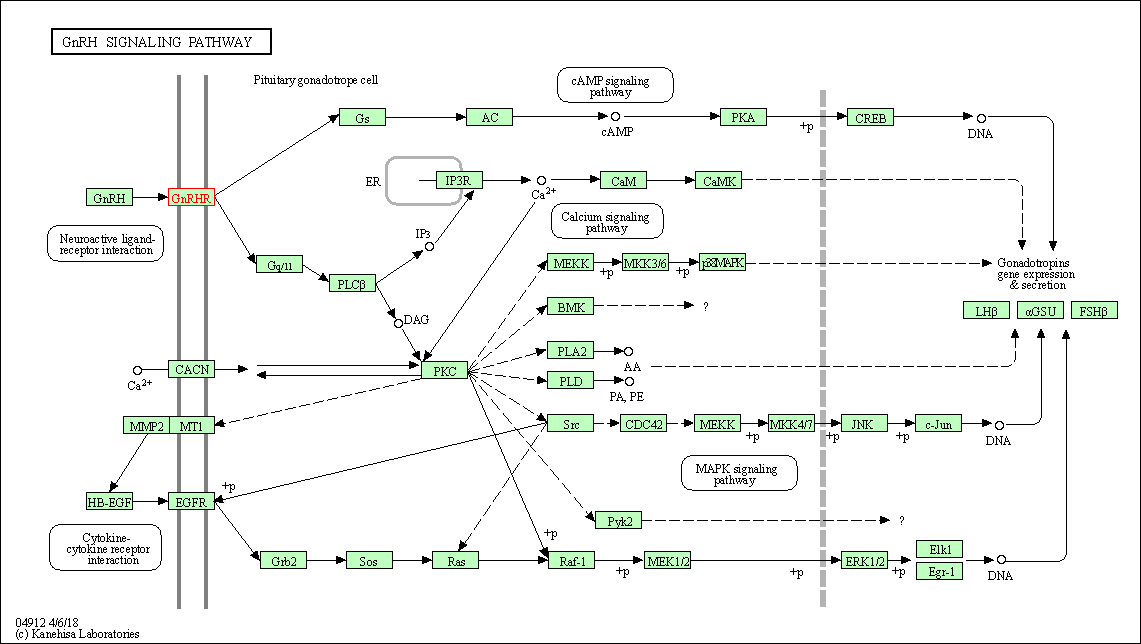
|
| Class: Organismal Systems => Endocrine system | Pathway Hierarchy | ||
| Degree | 3 | Degree centrality | 3.22E-04 | Betweenness centrality | 4.04E-04 |
|---|---|---|---|---|---|
| Closeness centrality | 1.86E-01 | Radiality | 1.31E+01 | Clustering coefficient | 3.33E-01 |
| Neighborhood connectivity | 1.90E+01 | Topological coefficient | 3.46E-01 | Eccentricity | 12 |
| Download | Click to Download the Full PPI Network of This Target | ||||
| Chemical Structure based Activity Landscape of Target | Top |
|---|---|
| Drug Property Profile of Target | Top | |
|---|---|---|
| (1) Molecular Weight (mw) based Drug Clustering | (2) Octanol/Water Partition Coefficient (xlogp) based Drug Clustering | |
|
|
||
| (3) Hydrogen Bond Donor Count (hbonddonor) based Drug Clustering | (4) Hydrogen Bond Acceptor Count (hbondacc) based Drug Clustering | |
|
|
||
| (5) Rotatable Bond Count (rotbonds) based Drug Clustering | (6) Topological Polar Surface Area (polararea) based Drug Clustering | |
|
|
||
| "RO5" indicates the cutoff set by lipinski's rule of five; "D123AB" colored in GREEN denotes the no violation of any cutoff in lipinski's rule of five; "D123AB" colored in PURPLE refers to the violation of only one cutoff in lipinski's rule of five; "D123AB" colored in BLACK represents the violation of more than one cutoffs in lipinski's rule of five | ||
| Target Poor or Non Binders | Top | |||||
|---|---|---|---|---|---|---|
| Target Poor or Non Binders | ||||||
| Target Regulators | Top | |||||
|---|---|---|---|---|---|---|
| Target-regulating Transcription Factors | ||||||
| Target Profiles in Patients | Top | |||||
|---|---|---|---|---|---|---|
| Target Expression Profile (TEP) | ||||||
| Target Affiliated Biological Pathways | Top | |||||
|---|---|---|---|---|---|---|
| KEGG Pathway | [+] 2 KEGG Pathways | + | ||||
| 1 | Neuroactive ligand-receptor interaction | |||||
| 2 | GnRH signaling pathway | |||||
| NetPath Pathway | [+] 2 NetPath Pathways | + | ||||
| 1 | IL1 Signaling Pathway | |||||
| 2 | IL2 Signaling Pathway | |||||
| Panther Pathway | [+] 1 Panther Pathways | + | ||||
| 1 | Heterotrimeric G-protein signaling pathway-Gq alpha and Go alpha mediated pathway | |||||
| Reactome | [+] 2 Reactome Pathways | + | ||||
| 1 | Hormone ligand-binding receptors | |||||
| 2 | G alpha (q) signalling events | |||||
| WikiPathways | [+] 5 WikiPathways | + | ||||
| 1 | Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| 2 | Peptide GPCRs | |||||
| 3 | GPCR ligand binding | |||||
| 4 | GPCR downstream signaling | |||||
| 5 | GPCRs, Other | |||||
| Target-Related Models and Studies | Top | |||||
|---|---|---|---|---|---|---|
| Target Validation | ||||||
| References | Top | |||||
|---|---|---|---|---|---|---|
| REF 1 | Gonadotropin releasing hormone analogs induce apoptosis by extrinsic pathway involving p53 phosphorylation in primary cell cultures of human prostatic adenocarcinomas. Prostate. 2009 Jul 1;69(10):1025-33. | |||||
| REF 2 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 5585). | |||||
| REF 3 | ClinicalTrials.gov (NCT01512472) Firmagon (Degarelix) Intermittent Therapy. U.S. National Institutes of Health. | |||||
| REF 4 | 2018 FDA drug approvals.Nat Rev Drug Discov. 2019 Feb;18(2):85-89. | |||||
| REF 5 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 1188). | |||||
| REF 6 | Abarelix for injectable suspension: first-in-class gonadotropin-releasing hormone antagonist for prostate cancer. Future Oncol. 2006 Dec;2(6):677-96. | |||||
| REF 7 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 1190). | |||||
| REF 8 | New drugs in development for the treatment of endometriosis. Expert Opin Investig Drugs. 2008 Aug;17(8):1187-202. | |||||
| REF 9 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 3877). | |||||
| REF 10 | Natural products as sources of new drugs over the last 25 years. J Nat Prod. 2007 Mar;70(3):461-77. | |||||
| REF 11 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 4391). | |||||
| REF 12 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | |||||
| REF 13 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 3902). | |||||
| REF 14 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health Human Services. 2020 | |||||
| REF 15 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 1177). | |||||
| REF 16 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 8362). | |||||
| REF 17 | ClinicalTrials.gov (NCT01620528) A Clinical Study to Evaluate the Safety and Efficacy of Elagolix in Subjects With Moderate to Severe Endometriosis-Associated Pain. U.S. National Institutes of Health. | |||||
| REF 18 | ClinicalTrials.gov (NCT01767155) Study Comparing AEZS-108/ZoptEC (Zoptarelin Doxorubicin in Endometrial Cancer) to Doxorubicin as a Second Line Therapy of Endometrial Cancer. U.S. National Institutesof Health. | |||||
| REF 19 | Clinical pipeline report, company report or official report of Ferring Pharmaceuticals Inc. | |||||
| REF 20 | ClinicalTrials.gov (NCT02147964) ITT-5 Mechanisms of Spermatogenesis. U.S. National Institutes of Health. | |||||
| REF 21 | ClinicalTrials.gov (NCT01767090) A Study to Assess the Effectiveness and Safety of Different Doses of ASP1707 Compared to Placebo for Endometriosis Associated Pelvic Pain. U.S. National Institutes ofHealth. | |||||
| REF 22 | ClinicalTrials.gov (NCT01533532) A Randomized, Placebo-controlled, Double-blind Study of KLH-2109 in Patients With Endometriosis (2). U.S. National Institutes of Health. | |||||
| REF 23 | Ozarelix, a fourth generation GnRH antagonist, induces apoptosis in hormone refractory androgen receptor negative prostate cancer cells modulating expression and activity of death receptors. Prostate. 2010 Sep 1;70(12):1340-9. | |||||
| REF 24 | ClinicalTrials.gov (NCT04843319) Open-Label, Proof-of-Concept and Dose Finding Phase 1b/2 Study of VERU-100 in Men With Advanced Prostate Cancer. U.S.National Institutes of Health. | |||||
| REF 25 | ClinicalTrials.gov (NCT00895466) Efficacy and Safety Study of the Therapeutic Vaccine PEP223 in Prostate Cancer Patients. U.S. National Institutes of Health. | |||||
| REF 26 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800007230) | |||||
| REF 27 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 3854). | |||||
| REF 28 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800000763) | |||||
| REF 29 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 1185). | |||||
| REF 30 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800017215) | |||||
| REF 31 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800003694) | |||||
| REF 32 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 1178). | |||||
| REF 33 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800013539) | |||||
| REF 34 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800014087) | |||||
| REF 35 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800008630) | |||||
| REF 36 | Coronaviruses - drug discovery and therapeutic options. Nat Rev Drug Discov. 2016 May;15(5):327-47. | |||||
| REF 37 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800003256) | |||||
| REF 38 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800013297) | |||||
| REF 39 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. | |||||
| REF 40 | Gonadotrophin-releasing hormone antagonists for assisted conception. Cochrane Database Syst Rev. 2006 Jul 19;3:CD001750. | |||||
| REF 41 | Age attenuates testosterone secretion driven by amplitude-varying pulses of recombinant human luteinizing hormone during acute gonadotrope inhibiti... J Clin Endocrinol Metab. 2007 Sep;92(9):3626-32. | |||||
| REF 42 | Use of cognitive behavior therapy for functional hypothalamic amenorrhea. Ann N Y Acad Sci. 2006 Dec;1092:114-29. | |||||
| REF 43 | Clinical pipeline report, company report or official report of Takeda (2009). | |||||
| REF 44 | Reversible downregulation of endocrine and germinative testicular function (hormonal castration) in the dog with the GnRH-agonist azagly-nafarelin ... Theriogenology. 2009 Apr 15;71(7):1037-45. | |||||
| REF 45 | Regulation of GnRH I receptor gene expression by the GnRH agonist triptorelin, estradiol, and progesterone in the gonadotroph-derived cell line alphaT3-1. Endocrine. 2006 Aug;30(1):139-44. | |||||
| REF 46 | Stability of cytotoxic luteinizing hormone-releasing hormone conjugate (AN-152) containing doxorubicin 14-O-hemiglutarate in mouse and human serum in vitro: implications for the design of preclinicalstudies. Proc Natl Acad Sci U S A. 2000 Jan 18;97(2):829-34. | |||||
| REF 47 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Target id: 256). | |||||
| REF 48 | Structure-activity relationship studies of gonadotropin-releasing hormone antagonists containing S-aryl/alkyl norcysteines and their oxidized deriv... J Med Chem. 2007 May 3;50(9):2067-77. | |||||
| REF 49 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800032657) | |||||
| REF 50 | Clinical pipeline report, company report or official report of Avarx. | |||||
| REF 51 | Antide (Nal-Lys GnRH antagonist) suppression of pituitary-testicular function and sexual behavior in group-living rhesus monkeys. Physiol Behav. 1991 Aug;50(2):429-35. | |||||
| REF 52 | Non-peptide gonadotropin-releasing hormone receptor antagonists. J Med Chem. 2008 Jun 26;51(12):3331-48. | |||||
| REF 53 | Physicochemical properties of A-75998, an antagonist of luteinizing hormone releasing hormone. J Pharm Sci. 1995 Aug;84(8):953-8. | |||||
| REF 54 | Discovery of sodium R-(+)-4-{2-[5-(2-fluoro-3-methoxyphenyl)-3-(2-fluoro-6-[trifluoromethyl]benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl]-1-phenylethylamino}butyrate (elagolix), a potent and orally available nonpeptide antagonist of the human gonadotropin-releasing hormone receptor. J Med Chem. 2008 Dec 11;51(23):7478-85. | |||||
| REF 55 | Effects of the luteinizing-hormone-releasing hormone (LHRH) antagonist ramorelix (hoe013) and the LHRH agonist buserelin on dimethylbenz[]anthracene-induced mammary carcinoma: studies with slow-release formulations. J Cancer Res Clin Oncol. 1993;119(8):457-62. | |||||
| REF 56 | Amiloride derivatives and a nonpeptidic antagonist bind at two distinct allosteric sites in the human gonadotropin-releasing hormone receptor. Mol Pharmacol. 2008 Jun;73(6):1808-15. | |||||
| REF 57 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800003256) | |||||
| REF 58 | Characterization of gonadotropin-releasing hormone analogs based on a sensitive cellular luciferase reporter gene assay. Anal Biochem. 1997 Aug 15;251(1):17-23. | |||||
| REF 59 | Inhibition of follicular development by a potent antagonistic analog of gonadotropin-releasing hormone (detirelix). J Clin Endocrinol Metab. 1991 Apr;72(4):927-33. | |||||
| REF 60 | Gonadotropin-releasing hormone receptor-targeted paclitaxel-degarelix conjugate: synthesis and in vitro evaluation.J Pept Sci.2015 Jul;21(7):569-76. | |||||
| REF 61 | Biological characterization of a novel, orally active small molecule gonadotropin-releasing hormone (GnRH) antagonist using castrated and intact rats. J Pharmacol Exp Ther. 2003 May;305(2):688-95. | |||||
| REF 62 | Rescue of hypogonadotropic hypogonadism-causing and manufactured GnRH receptor mutants by a specific protein-folding template: misrouted proteins as a novel disease etiology and therapeutic target. JClin Endocrinol Metab. 2002 Jul;87(7):3255-62. | |||||
| REF 63 | In vivo characterization of a novel GnRH (gonadotropin-releasing hormone) antagonist, LXT-101, in normal male rats. Regul Pept. 2006 Sep 11;136(1-3):122-9. | |||||
| REF 64 | In vivo effects of a potent GnRH antagonist ORG 30850: physiologic evidence that down-regulation of GnRH receptors does not occur. J Soc Gynecol Investig. 1994 Oct-Dec;1(4):290-6. | |||||
| REF 65 | Overlapping, nonidentical binding sites of different classes of nonpeptide antagonists for the human gonadotropin-releasing hormone receptor. J Med Chem. 2006 Jan 26;49(2):637-47. | |||||
| REF 66 | Synthesis and biological evaluation of piperazinyl heterocyclic antagonists of the gonadotropin releasing hormone (GnRH) receptor. Bioorg Med Chem Lett. 2010 Apr 15;20(8):2512-5. | |||||
| REF 67 | Residues within transmembrane helices 2 and 5 of the human gonadotropin-releasing hormone receptor contribute to agonist and antagonist binding. Mol Endocrinol. 2000 Jul;14(7):1099-115. | |||||
| REF 68 | Novel analogues of degarelix incorporating hydroxy-, methoxy-, and pegylated-urea moieties at positions 3, 5, 6 and the N-terminus. Part III. J Med Chem. 2006 Jun 15;49(12):3536-43. | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

