Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D09AOO
|
||||
| Former ID |
DCL001141
|
||||
| Drug Name |
ANAVEX 2-73
|
||||
| Company |
Anavex
|
||||
| Structure |
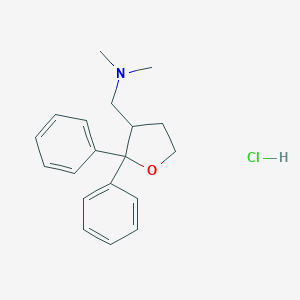
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | Sigma(1)-type opioid receptor | Target Info | Agonist | [549727] | |
| References | |||||
| Ref 531153 | Anti-amnesic and neuroprotective potentials of the mixed muscarinic receptor/sigma 1 (?1) ligand ANAVEX2-73, a novel aminotetrahydrofuran derivative. J Psychopharmacol. 2011 Aug;25(8):1101-17. | ||||
| Ref 532266 | Blockade of Tau hyperphosphorylation and Abeta?????? generation by the aminotetrahydrofuran derivative ANAVEX2-73, a mixed muscarinic and ???receptor agonist, in a nontransgenic mouse model of Alzheimer's disease. Neuropsychopharmacology. 2013 Aug;38(9):1706-23. | ||||
| Ref 549727 | 2011 Pipeline of Anavex. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

