Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D09VMI
|
||||
| Former ID |
DCL000354
|
||||
| Drug Name |
Cediranib
|
||||
| Synonyms |
Recentin; AZD 2171; AZD2171; M6294; AZD-2171; ZD-2171; AZD2171, Recentin, Cediranib; Cediranib (USAN/INN); 4-[(4-fluoro-2-methyl-1H-indol-5-yl)oxy]-6-methoxy-7-(3-pyrrolidin-1-ylpropoxy)quinazoline; 4-[(4-fluoro-2-methyl-1H-indol-5-yl)oxy]-6-methoxy-7-[3-(pyrrolidin-1-yl)propoxy]quinazoline
|
||||
| Drug Type |
Small molecular drug
|
||||
| Therapeutic Class |
Anticancer Agents
|
||||
| Company |
AstraZeneca
|
||||
| Structure |
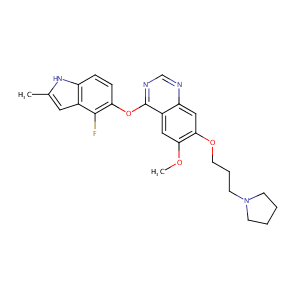
|
Download2D MOL |
|||
| Formula |
C25H27FN4O3
|
||||
| InChI |
InChI=1S/C25H27FN4O3/c1-16-12-17-19(29-16)6-7-21(24(17)26)33-25-18-13-22(31-2)23(14-20(18)27-15-28-25)32-11-5-10-30-8-3-4-9-30/h6-7,12-15,29H,3-5,8-11H2,1-2H3
|
||||
| InChIKey |
XXJWYDDUDKYVKI-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 288383-20-0
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
14906490, 24208254, 26746631, 57373832, 75977042, 96025564, 96079499, 99432367, 103592924, 103905340, 109692967, 123110211, 124756936, 124766796, 125163743, 126666991, 126730931, 129754723, 131465106, 134221888, 134339006, 134964400, 135194712, 135697662, 135727476, 136368003, 136920391, 137006068, 137275941, 139208216, 144115802, 152258121, 152344142, 160646960, 162011780, 162037386, 162200059, 163884606, 164041832, 174528685, 175426860, 177748726, 178102292, 180190844, 185998790, 187071991, 198958766, 198993121, 203355740, 223384883
|
||||
| Target and Pathway | |||||
| Target(s) | Vascular endothelial growth factor receptor 2 | Target Info | Inhibitor | [536474], [550288] | |
| NetPath Pathway | IL2 Signaling Pathway | ||||
| PANTHER Pathway | Angiogenesis | ||||
| VEGF signaling pathway | |||||
| Pathway Interaction Database | HIF-2-alpha transcription factor network | ||||
| Beta3 integrin cell surface interactions | |||||
| Signaling events mediated by TCPTP | |||||
| SHP2 signaling | |||||
| S1P1 pathway | |||||
| VEGF and VEGFR signaling network | |||||
| Integrins in angiogenesis | |||||
| Signaling events mediated by VEGFR1 and VEGFR2 | |||||
| Notch-mediated HES/HEY network | |||||
| References | |||||
| Ref 525194 | ClinicalTrials.gov (NCT02446600) Olaparib or Cediranib Maleate and Olaparib Compared With Standard Platinum-Based Chemotherapy in Treating Patients With Recurrent Platinum-Sensitive Ovarian, Fallopian Tube, or Primary Peritoneal Cancer. U.S. National Institutes of Health. | ||||
| Ref 541007 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 5664). | ||||
| Ref 536474 | A comparison of physicochemical property profiles of marketed oral drugs and orally bioavailable anti-cancer protein kinase inhibitors in clinical development. Curr Top Med Chem. 2007;7(14):1408-22. | ||||
| Ref 550288 | Clinical pipeline report, company report or official report of AstraZeneca (2009). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

